Marks told reporters it will likely take at least another couple of months before human data on the BA.5 boosters is available to the public. When Pfizer's and Moderna's shots were authorized in December 2020, they provided more than 90% protection at preventing Covid. The FDA will conduct surveillance to see whether the boosters meet that goal, Marks said. And that is what we believe the evidence that we've seen helps point to," Marks told reporters during a news conference after the FDA authorization Wednesday. "We don't know for a fact yet whether we will get to that same level, but that is the goal here. But Marks acknowledged that the federal government's experts simply do not know yet whether the boosters will meet the high bar set by those doses. Peter Marks, who heads the FDA office that reviews vaccines, said the new boosters aim to restore the high levels of protection that vaccines demonstrated in early 2021. They are worried that the waning effectiveness of the old vaccines is creating an opening for omicron to cause another wave of hospitalizations this winter as people spend more time indoors where the airborne virus spreads more easily.ĭeaths and hospitalizations have climbed since April among the elderly, the most vaccinated age group in America, as omicron has continued to mutate into more and more transmissible subvariants that dodge the protection of the original vaccines, according to Heather Scobie, a CDC epidemiologist.ĭr. The nation's top health officials acted with urgency this summer to ensure the new boosters would roll out in time for the fall. The shots also target the original strain of the virus that first emerged in Wuhan, China, in 2019. 
The new boosters, authorized for people ages 12 and older, target the highly contagious and immune-evasive omicron BA.5 subvariant that has caused a wave of breakthrough infections over the summer. Personal Loans for 670 Credit Score or Lower Personal Loans for 580 Credit Score or Lower 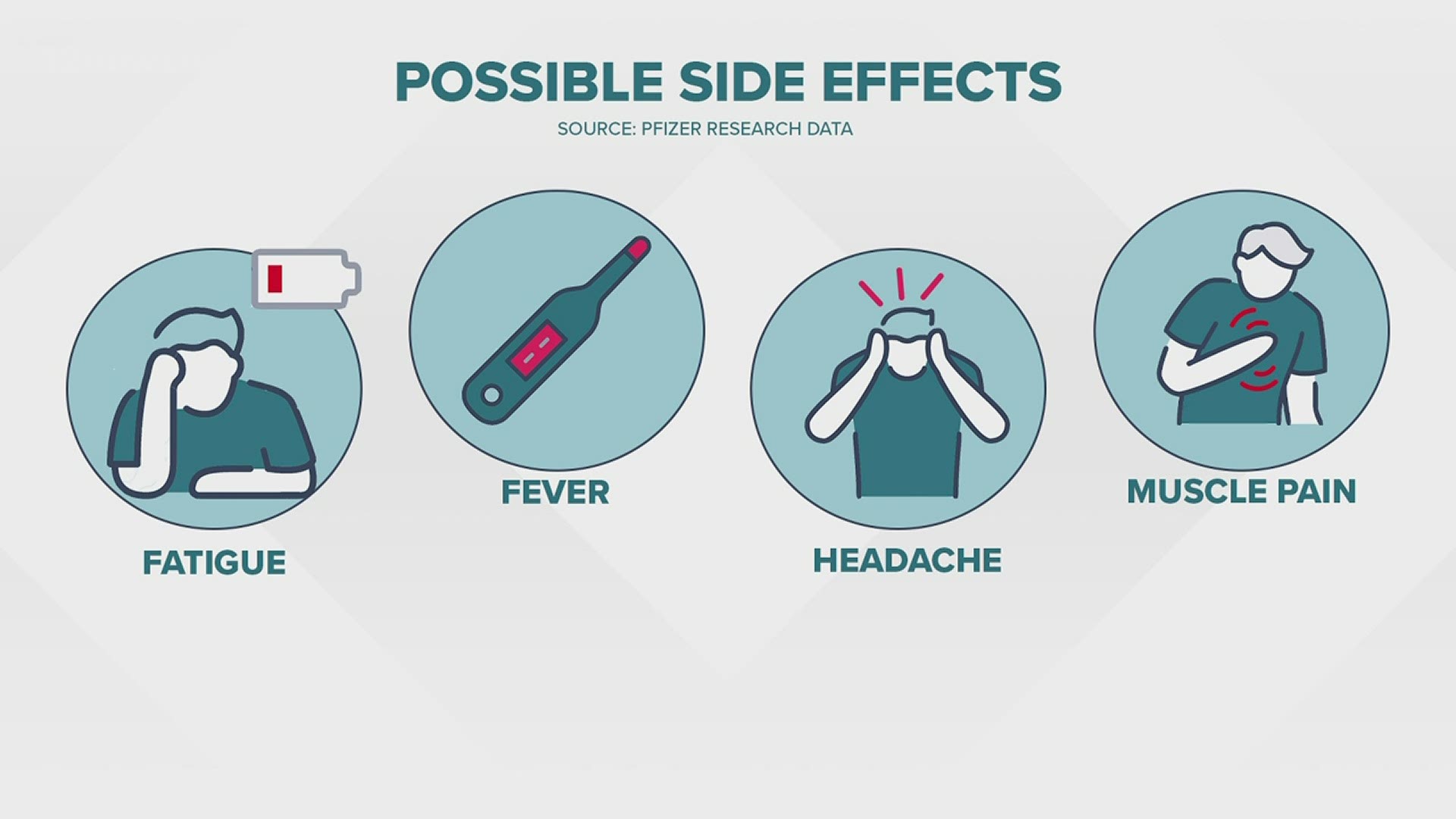
"This booster is likely going to give you better protection in the fall," Cooke said in a recent interview.Best Debt Consolidation Loans for Bad Credit John Cooke, the director of the Center for RNA Therapeutics at Houston Methodist. The new booster is especially recommended for those who are higher risk for developing severe disease, such as anyone over the age of 65 or with a preexisting medical condition, said Dr. Centers for Disease Control and Prevention data shows BA.4 and BA.5 still account for more than 95 percent of the virus circulating nationwide. MORE HEALTH NEWS: Harris County's new COVID threat levels will focus on new cases, not hospitalizationsĭoctors recommend that anyone who is eligible should get the new booster, particularly because U.S. The new booster can be given two months after someone received their last booster or completed their initial vaccination series, officials said. If you’ve already been vaccinated or boosted, you do not need to stick with the same manufacturer for the new dose, officials said. The other, made by Moderna, is available to those 18 and older. 
One new version, made by Pfizer/BioNTech, is available to anyone 12 and older. The new boosters still offer protection against the original strain of COVID-19, but they also protect against omicron and its BA.4 and BA.5 subvariants. “There’s not anything there that would suggest we should see a very different side effect profile,” Chang said in a recent interview.įederal health officials approved the updated boosters earlier this month. The one notable difference is that the updated boosters also contain genetic coding for the spike protein of omicron and its BA.4 and BA.5 subvariants. That’s because the components of the updated boosters are essentially the same as those in the original vaccines. Michael Chang, a pediatric infectious disease specialist at UTHealth Houston and Children’s Memorial Hermann Hospital. The fact that the most common side effects are largely unchanged is not surprising, said Dr. The Pfizer study involved approximately 600 participants, while the Modern study involved approximately 800, according to the Food and Drug Administration.Įxperts have noted that side effects can be a good sign, because they indicate that the immune system is responding to the vaccine. Participants in the Moderna trial also reported swelling of the lymph nodes in the same arm of the injection, as well as nausea and vomiting. MORE HEALTH NEWS: Houston doctors say flu season could be early and more severe, so get a flu shot now They included pain, redness and swelling at the injection site fatigue headache muscle pain chills joint pain and fever. Clinical studies that evaluated the safety of the boosters made by Pfizer/BioNTech and Moderna found that each was associated with many of the same side effects as the original vaccines. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
